Clinical trials are research studies conducted with people affected by HAE to evaluate whether a drug is safe and effective in humans. They are one of the final stages of the medical research and development process and follow strict standards to protect people. Clinical trials for investigational drugs typically go through 3 phases before the FDA reviews and approves them. Sometimes drugs enter into a Phase 4 study, also called a post-approval study, that is offered to take the approved medication to continue to monitor the safety and efficacy of the drug.
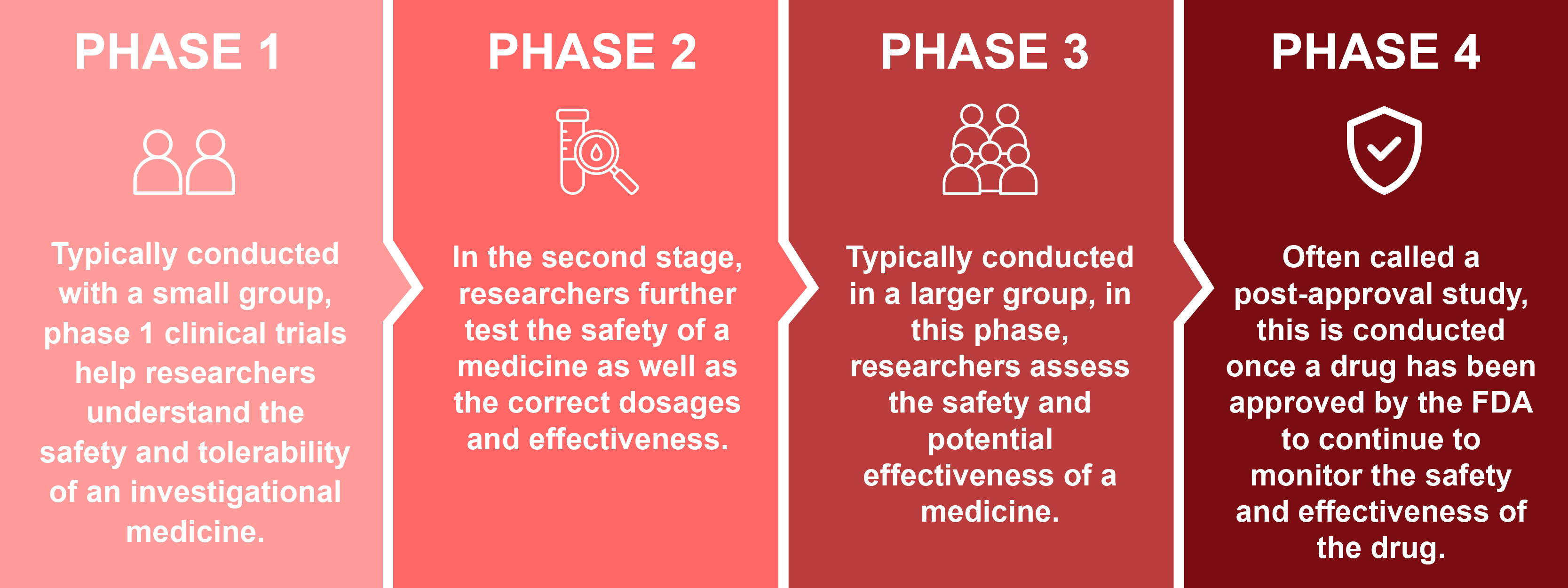
Clinical trials are a critical part of drug development and are required before a treatment can be approved by the Federal Drug Administration (FDA) and become available by prescription.
What are the benefits of participating in a Clinical Trial?
Participating in a clinical trial offers several important benefits. You have the opportunity to make a meaningful contribution to the future of HAE treatments, gain access to potential new HAE medications, and receive close medical supervision from a knowledgeable HAE physician. In addition, participants may be compensated for their time and involvement in the study.
Please return to this page often to check for clinical trial updates. Contact any of the HAE Advocates to learn about the eight FDA-approved HAE therapies currently available that can transform the lives of people with HAE








If you are interested in participating in a clinical trial, please click below to contact Christine Selva to see if there is an active clinical trial site in your area.
Ensure you include the following information in your submission: